The Future of Research: Studying Human Organs and Diseases on a Chip
Cedars-Sinai Investigators Extol New Research Models, Describe How to Create a 3D Heart
Replicating human organs using stem cells and lab-on-a-chip technology can revolutionize the way diseases and potential treatments are studied, according to two new papers by Cedars-Sinai investigators.
Stem cells are cells that have the potential to develop into many different cell types. A lab-on-a-chip is a miniaturized laboratory system that can be used to create a multidimensional organ model.
“Cedars-Sinai is perfectly positioned to apply these technologies, because we’re both a basic science institute and a healthcare provider, so we can validate and clinically test them,” said Arun Sharma, PhD, a stem cell biologist in the Board of Governors Regenerative Medicine Institute, Department of Biomedical Sciences, Cancer Institute and Smidt Heart Institute at Cedars-Sinai. Sharma is senior and corresponding author on both of the new publications.
Sharma and colleagues describe in a paper published in the journal Current Protocols how they have created a 3D model using beating clumps of heart tissue. This technique is an improvement upon 2D heart tissue models the investigators have been using to study the heart since 2020.
Both types of models include heart cells derived from human induced pluripotent stem cells (iPSCs). These are stems cells that are created in a laboratory from a person’s blood and skin cells. The cells are then engineered to resemble any cell in the body.
For their 2D heart model, for example, investigators turned iPSCs into beating heart cells called cardiomyocytes. In one study, they exposed the cells to the virus that causes COVID-19, which led them to discover that the virus can infect the heart directly.
The 3D model comprises cardiomyocytes as well as other cells found in the heart, such as fibroblasts, connective tissue cells and endothelial cells, which line the blood vessels in the heart.
The cells are cultured in petri dishes and then placed together in a laboratory plate, forming a sphere. The miniature sphere used to create these multidimensional organs is known as a cardiac spheroid. The resulting spheroid can be used to study the biology of the heart and how it is affected by various drugs or infections.
The protocols paper by Sharma and colleagues is meant to serve as a guide for other investigators interested in replicating the process of making a 3D heart model to study heart biology.
Cedars-Sinai investigators are also currently using 3D heart models to study how cancer drugs might affect the heart.
“We have multiple types of cells in these 3D clusters, so we can predict which cells are going to be the most susceptible to damage from chemotherapy drugs,” Sharma said.
Cedars-Sinai investigators Madelyn Arzt, Stephany Pohlman and Maedeh Mozneb, PhD, also authored the paper.
The work by Sharma and colleagues was inspired by other Cedars-Sinai scientists. Under the guidance of Clive Svendsen, PhD, professor of Biomedical Sciences and Medicine and executive director of the Board of Governors Regenerative Medicine Institute at Cedars-Sinai, investigators have created brain-on-a-chip technology using patients’ own cells.
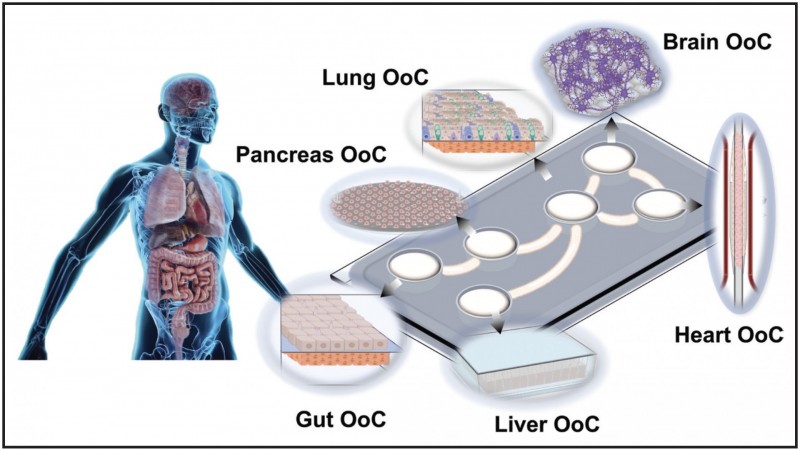 In another paper, Sharma, Arzt and Mozneb describe how recent advances in stem cell and lab-on-a-chip technology have improved understanding of COVID-19. The review paper was published in Circulation Research.
In another paper, Sharma, Arzt and Mozneb describe how recent advances in stem cell and lab-on-a-chip technology have improved understanding of COVID-19. The review paper was published in Circulation Research.
Investigators have been converting stem cells into cells that are similar to those found on tissues and organs in the human body and then using them to study how the virus that causes COVID-19 damages cells.
“Most excitingly, we can use these models to identify new drugs that can either kill the virus itself or alleviate some of the cardiac symptoms that people would experience as part of COVID-19,” Sharma said.
The potential for these research models is unlimited, according to Sharma and colleagues.
“You can think of any disease type, and you can probably use these types of models to study that disease,” Sharma said.
Funding: The paper published in Current Protocols was supported by the American Heart Association (Career Development Award 856987), the Board of Governors Regenerative Medicine Institute at Cedars-Sinai, an In-Space Production Award (InSPA) from NASA, the Donna and Jesse Garber Award for Cancer Research, and a California Institute for Regenerative Medicine (CIRM) Bridges Award.
The paper published in Circulation Research was supported by the American Heart Association (Career Development Award 856987, COVID-19 Rapid Response Grants 814630 and 20203858), the Board of Governors Regenerative Medicine Institute at Cedars-Sinai, the Donna and Jesse Garber Award for Cancer Research, the National Institutes of Health (award numbers HL.118650, HL149808, HL129727, HL159970, HL165318), and Veterans Affairs Merit Award I01 BX004356.
Follow Cedars-Sinai Academic Medicine on Twitter for more on the latest basic science and clinical research from Cedars-Sinai.




