Research Areas
The Van Eyk Laboratory is fortunate to have many great collaborators with whom we develop, sustain and share a diverse array of research projects. We also work closely with our industry partners to bring the latest technologies to bear on our clinically driven research. Our group comprises a discovery-based lab and a high-throughput facility. These twin components operate in close concert within the Advanced Clinical Biosystems Research Institute.
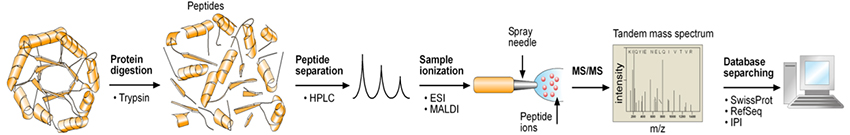
"Bottom-up" proteomic analysis. 1. Proteins in a sample are digested with trypsin to form peptides. 2. Peptides are analytically separated, ionized, and introduced to a mass spectrometer. 3. Peptides are sequentially analyzed, fragmented, and reanalyzed as peptide fragments in the mass spectrometer in a process called "tandem mass spectrometry" or "MS/MS" analysis. 4. MS/MS results are searched against established and predicted databases to reveal the identities of the proteins present in the original sample.
Maladaptive Proteomic Signaling in Aortic Aneurysm and Transforming Growth Factor Beta (TGFβ) Vasculopathies
Aortic aneurysm is a common clinical manifestation of several TGFβ vasculopathies including Marfan and Loeys-Dietz syndromes. This prevalent condition is defined by excessive aortic growth and medial wall remodeling that can result in lethal dissection and rupture. Few effective pharmacological treatments exist for aneurysm, largely due to an incomplete understanding of the disease mechanism. Preliminary in vitro and in vivo data indicate that blockading the angiotensin II type 1 receptor (AT1R) using losartan potently inhibits aneurysm progression in mouse models. While the effective dose in mice exceeds the approved dose for treatment of children and adolescents, these promising animal results may provide proteomic clues that reveal mechanistic and potentially therapeutic targets. Research in the Van Eyk Laboratory aims to delineate the proteomic signatures of losartan therapy, downstream of AT1R in mouse vascular smooth muscle cells, resulting in a protective phenotype. Our goal is to identify signaling pathways and intermediates that serve as additional candidate therapeutic targets.
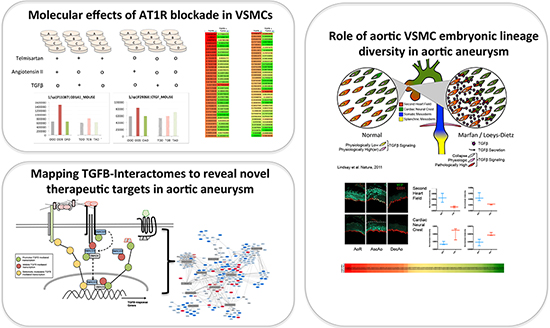
| Our proteomic experiments into the basic mechanisms underlying aortic aneurysm are guided by clinical observations in human disease. In some of our experiments, we are attempting to better understand the anatomical specificity of aortic aneurysm by focusing on subtle molecular differences between smooth muscle cells in the aorta that are derived from different embryonic lineages (right panel). In other work, we are integrating knowledge from the clinic as to cell signaling pathways that intersect to drive aneurysm progression and mapping points of convergence between them (left panels). We intend to utilize the knowledge gained from these and other basic molecular experiments to validate potential targets in vivo, and ultimately move novel therapeutic strategies toward clinical implementation |
Proteomics in Cardiac Resynchronization Therapy for Heart Failure
Cyclic-guanosine monophosphate (cGMP) signaling is increasingly recognized as playing an important role in heart failure. The Van Eyk Lab works to better understand both pathophysiological signaling in heart failure and mechanistic signaling of cardiac resynchronization therapy. Ron Holewinski, PhD, was part of the team of researchers headed by David Kass, MD, at Johns Hopkins University that showed that cGMP arising from either nitric oxide or natriuretic peptide pathways is hydrolyzed by specific phosphodiesterases. This team also showed that the therapeutic effects of cardiac resynchronization therapy might be mediated by a reactivation of glycogen synthase kinase 3 beta.
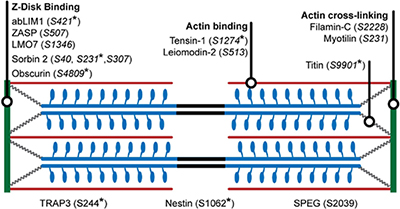
| Research in the Van Eyk Laboratory has identified a host of new phosphorylation events on several proteins in the sarcomere. Several of these have been implicated with Cardiac Resynchronization Therapy. |
Protein Citrullination in Cardiac, Neurodegenerative and Autoimmune Diseases
The Van Eyk Laboratory is interested in protein deimination (citrullination) and the role that this process plays in heart diseases including ischemic, dilated cardiomyopathy. This form of posttranslational protein modification creates novel epitopes on common proteins, providing "neoantigens" that are now known to be important in autoimmune disease. This research is a major focus of Justyna Fert-Bober, PhD, who hypothesizes that citrullination plays a key role in the molecular mechanism that drives heart disease in patients with rheumatoid arthritis, which, together with many other important autoimmune diseases, disproportionately affects women. Other citrullination events involve the pathophysiology of traumatic brain injury, and contribute to protein degradation and neurodegeneration. In this sense, citrullinated proteins may facilitate the discovery of new biomarkers that could improve diagnostic and prognostic standards in clinical use.
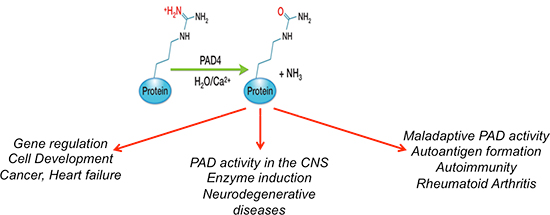
| Citrullination, or deimination, occurs when arginine residues on proteins are converted to citrulline through the activity of peptidylarginine deiminase (PAD), left panel. PAD activation can a) alter protein function and contribute to disease pathogenesis, b) result in biomarkers that reflect disease states, and c) produce autoantigens that contribute to autoimmune disorders. |
Biomarker Discovery and Validation — The WISE Study
The Van Eyk Laboratory will play a role in the Women's Ischemic Syndrome Evaluation (WISE) study. This landmark study, led by C. Noel Bairey Merz, MD, seeks new insights into those factors that distinguish heart disease in women, in an effort to inform standards of care and ultimately improve outcomes. The Van Eyk Lab is looking to bring our expertise to bear on several aspects within this larger study. Specifically, our ability to perform ultrasensitive assays for cardiac troponin, among other biomolecules, while simultaneously studying its disease-specific variants, will endow the WISE study with additional biochemical parameters with which to distinguish and potentially diagnose ischemic heart disease in women for whom no sufficient tests currently exist.
Programs of Excellence in Glycosciences — Glycobiology
The biological roles of glycans are often overlooked, but their fundamental importance is underscored by the fact that complex and dense surface glycosylation is a universal feature of all living cells. Glycans attached to proteins are not decorative; they contribute to proper protein function. Extracellular glycans can modulate receptor sensitivity and protein configuration, while intracellular O-GlcNAcylation is a dynamic posttranslational modification involved in signaling. It is in these contexts that the Van Eyk Lab — as part of the glycoconjugates and cardiovascular disease branch within the National Heart, Lung and Blood Institute Programs of Excellence in Glycosciences — is interested in glycobiology.
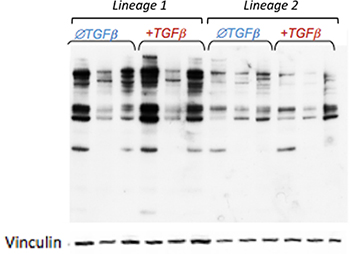
The extent and pattern of dynamic O-GlcNAcylation on intracellular proteins differs in vascular smooth muscle cells derived from different lineages upon TGFβ-1 stimulation. |
Contact the Van Eyk Lab
127 S. San Vicente Blvd.
Pavilion, 9th Floor
Los Angeles, CA 90048
