Atherosclerosis
Preclinical Research
To facilitate deeper understanding of atherosclerosis and plaque instability, Cedars-Sinai principal investigators are studying the role of novel genes and identify molecular pathways in genetically engineered mice. More than 15 studies are currently being conducted, including:
- Understanding the role of the immune system in atherosclerosis using animal models. Cedars-Sinai studies show that an autoimmune response to antigens within the protein component of low-density lipoprotein cholesterol stimulates the innate and adaptive immune system, which perpetuates vascular inflammation and contributes to the buildup of artery-clogging cholesterol plaque. To reduce inflammation and plaque progression, researchers are:
- Developing novel methods to modulate the immune system, such as vaccines created from apo 100 protein fragments
- Principal investigators: P.K. Shah, MD, Kuang-Yuh Chyu, MD, PhD, and Paul Dimayuga, MD
- Using animal models to test the potential benefits of the apoB-100-related vaccine in cholesterol buildup, autoimmune response and inflammation, high blood pressure and aortic aneurysms. Preliminary results suggest that the vaccine reduces plaque buildup and also reduces high blood pressure and death from aortic aneurysm rupture.
- Principal investigators: P.K. Shah, MD, and Kuang-Yuh Chyu, MD, PhD
- Principal investigators: P.K. Shah, MD, and Kuang-Yuh Chyu, MD, PhD
- Investigating the immune proteome of coronary artery disease patients to identify potentially new autoantigens, which may play a role in atherosclerosis. The self-antigens identified are being tested in animal models to modulate the immune response to atherosclerosis.
- Principal Investigators: Paul Dimayuga, PhD, and Bojan Cercek, MD, PhD
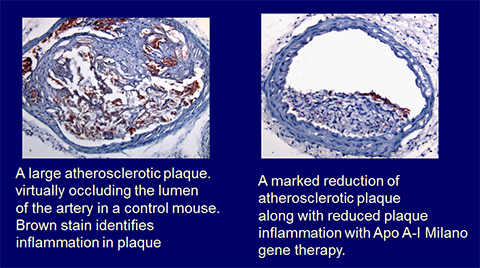
- Exploiting the benefits and effects of mutant protein apoA-1 Milano for prevention and reversal of plaque buildup. Researchers have developed the concept of using infusions of apoA-1 Milano — a naturally occurring protein produced by a mutant gene carried by a few individuals in Limone sul Garda, Italy — to prevent and reverse atherosclerosis. Cedars-Sinai studies in animal models reveal that transferring this gene using AAV vectors, an innocuous virus, as a carrier rapidly reverses cholesterol plaque buildup. Investigators are pursuing plans to test this gene-therapy approach in humans. Studies of protein apoA-1 Milano infusions in humans are underway in Europe.
- Principal investigator: P.K. Shah, MD
- Evaluating the role of novel genes tenascin-C and pleiotrophin in arterial blockage and cardiomyopathy using transgenic mouse models to identify new molecular targets for treatment and prevention of occlusive vascular disease and cardiomyopathy.
- Principal investigators: P.K. Shah, MD, and Behrooz Sharifi, PhD
- Principal investigators: P.K. Shah, MD, and Behrooz Sharifi, PhD
- Examining the transcriptional mechanisms and GATA-3 gene that regulate the behavior of macrophage subtypes: classically activated M1 (pro-inflammatory) versus alternatively activated M2 (anti-inflammatory). The ultimate goal is to identify new molecular targets for modifying the pro-inflammatory cells into anti-inflammatory phenotypes for managing chronic inflammatory disease, including atherosclerosis.
- Principal Investigators: P.K. Shah, MD, and Behrooz Sharifi, PhD
- Principal Investigators: P.K. Shah, MD, and Behrooz Sharifi, PhD
- Investigating the metabolic and cardiovascular phenotype of mice lacking the KLF14 gene, which appears to be a master regulator of fat and possibly glucose metabolism, using KLF14 knockout mice. These studies are designed to provide new knowledge that may identify novel targets for obesity and diabetes/metabolic syndrome management.
- Principal Investigators: P.K. Shah, MD, and Behrooz Sharifi, PhD
- Investigating the potential benefits of gene transfer, using an innocuous virus called AAV9 to deliver the apo A-1 Milano gene, in mouse models of Alzheimer’s dementia.
- Principal Investigators: P.K. Shah, MD, and Behrooz Sharifi, PhD, in collaboration with Maya Koronyo, PhD
Clinical Research
Cedars-Sinai clinical research includes two multicenter trials focusing on non-statin, cholesterol-lowering pharmaceutical therapies:
- PCSK9 antibodies, a class of non-statin, cholesterol-lowering medication, targeting the proprotein convertase subtilisin/kexin type 9 gene. These drugs block PCSK9 actions and can lower LDL cholesterol by 50 to 70 percent with subcutaneous injections once every two to four weeks.
- Principal investigator: P.K. Shah, MD
- Principal investigator: P.K. Shah, MD
- Lomitapide (Juxtapid), a microsomal triglyceride transfer protein inhibitor, to treat homozygous familial hypercholesterolemia, a rare genetic disorder that causes abnormally high LDL cholesterol levels and puts patients at risk for premature atherosclerosis and death. Now entering the registry phase, this trial has already resulted in the U.S. Food and Drug Administration approving lomitapide. Homozygous familial hypercholesterolemia patients now have an alternative to the only treatment previously available — LDL apheresis, a dialysis-like process in which a machine filters the patient’s blood to remove LDL
- Principal investigator: P.K. Shah, MD
- Principal investigator: P.K. Shah, MD
Have Questions or Need Help?
Contact us if you have questions or wish to learn more about the programs and services at the Smidt Heart Institute at Cedars-Sinai.
