Research Areas
Researchers in the Saghizadeh Ghiam Laboratory study the microRNA regulatory mechanisms of limbal epithelial stem cell (LESC) proliferation, migration, adhesion and differentiation in normal cornea homeostasis and wound-healing processes. Understanding these mechanisms could be key to understanding corneal diseases. In addition, the Saghizadeh Ghiam Lab studies the contribution of limbal extracellular vesicles (EVs) derived from corneal epithelial (LECs) and stromal cells in the molecular processes that lead to regeneration and wound healing.
Treating Corneal Diseases
A wide variety of physiological and pathological conditions, such as infections, inflammations and corneal diseases including limbal stem cell deficiency (LSCD) and diabetes can affect LESC functions, resulting in serious vision problems. Altered wound-healing processes, as seen in diabetic cornea, can result in corneal vascularization and loss of transparency, leading to corneal blindness. Currently, there is a need to better understand the mechanisms responsible for these abnormalities and develop new and efficacious treatments. The mechanisms of LESC proliferation, migration, adhesion and differentiation in normal corneal homeostasis and wound healing could be key to managing corneal diseases. Therefore, the understanding of miRNA and EVs regulatory mechanisms is critically important in the etiology and treatment of various corneal diseases such as LSCD and diabetic keratopathy, which are the major causes for corneal blindness.
Understanding the Role of MicroRNAs
The Saghizadeh Ghiam Laboratory employs a variety of molecular, functional tests and ex vivo human organ-cultured corneas to understand the role of microRNAs in both differentiated epithelial and stem cells of normal and diabetic corneas and their roles in driving LESC activation in normal and diseased cornea. The focus of the lab is determining the targets of microRNAs, and attempting gene therapy with either overexpression or silencing of specific microRNAs to normalize corneal wound healing and LESC marker patterns in diabetic cornea. The lab targets diabetic corneal epithelial cells by single microRNA, or their combinations, to improve function of corneal epithelium. This research could lead to the generation of a new cell source for restoring vision in patients with altered LESC.
The Saghizadeh Ghiam Lab has identified several miRNAs with altered expression in diabetic corneas using gene array, which correlates with their effects on corneal cell wound healing. The lab has also performed next generation sequencing (NGS) analysis of a large group of human normal and diabetic corneas resulting in the identification of promising miRNA candidates for further analysis. These studies may lead to restoration of normal functions to diabetic corneal stem cells. The Saghizadeh Ghiam Laboratory has successfully treated delayed wound healing in human diabetic organ-cultured corneas by targeting miR-146a in human diabetic organ-cultured corneas.
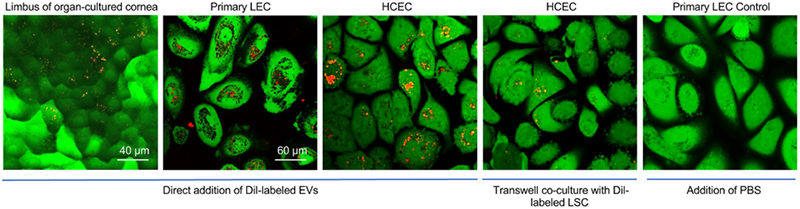
Understanding the Role of Extracellular Vesicles
The important part of the limbal stem cell niche functioning is interaction between stem cells and their neighboring stromal cells through secreted factors such as nanosized extracellular vesicles (EVs). These vesicles contain mRNA, miRNA, DNA and protein cargo mediating physiological intercellular crosstalk. The Saghizadeh Ghiam Laboratory investigates limbal EV contribution to corneal regeneration and wound healing, and examines possible differences in EV cargos released from normal and diabetic limbal stromal cells (LSC) and LECs by using different techniques such as EV isolation, miRNA isolation, next-generation sequencing, proteomics, transmission electron and confocal microscopy, and trans-well co-culture system. The laboratory shows that normal LSC-derived EVs significantly promote epithelial healing in wounded organ-cultured human corneas. In addition, the lab found that there is a difference in cargos of EVs derived from normal and diabetic LSC.
Investigation of molecular changes in diabetic corneas is essential for understanding of underlying causes and identification of therapeutic targets for diseased cornea and wound healing process in different layers of the cornea.
Contact the Saghizadeh Ghiam Lab
Advanced Health Sciences Pavilion, Suite A8104
127 S. San Vicente Blvd.
Los Angeles, CA 90048
