October 2019 Case
Authors
Elias Makhoul DO (PGY-3); Fabiola Medeiros MD (Faculty)
Subject: Placental Pathology
Clinical History
34-year old G3P0 with a prior history significant for chronic inflammatory demyelinating polyneuropathy status post immunosuppressive therapy with an estimated gestational age by LMP of 31 weeks and 4 days when she experienced no fetal movement. Ultrasonography confirmed lack of cardiac activity, fetal demise, and demonstrated severe intrauterine growth restriction. A fetopsy with cytogenetic analysis was requested to evaluate for etiology of fetal demise.
Figures
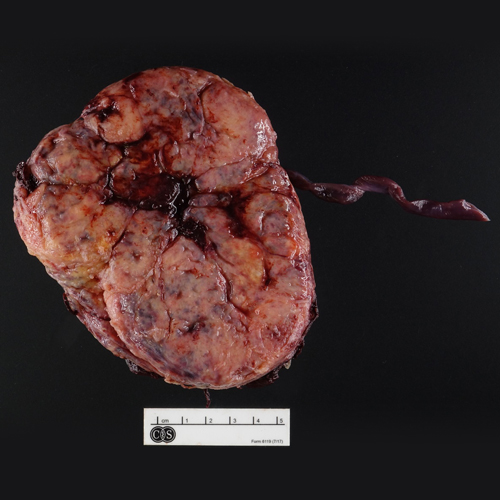
Figure 1 (Gross Photo, Maternal Surface) Gross photo shows a diffusely pale yellow-tan maternal surface with effacement of cotyledons.
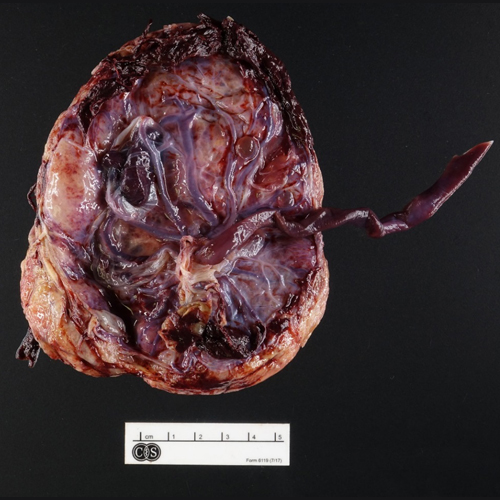
Figure 2 (Gross Photo, Fetal Surface) Gross photo shows fetal surface with diffuse subchorionic fibrin/fibrinoid deposition
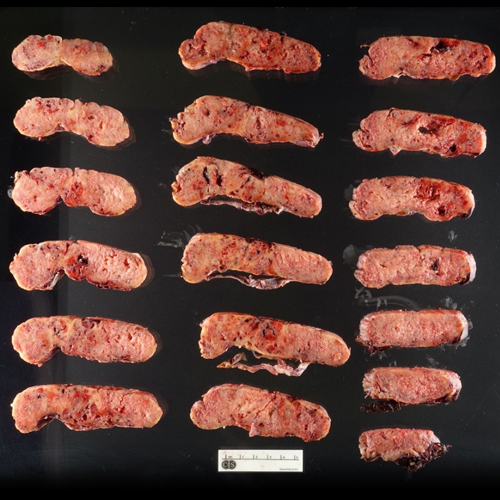
Figure 3 (Gross Photo, Cross Section) Serial sections show consolidation of the parenchyma with deposition of fibrin/fibrinoid material extending upward towards the chorionic plate in a matted pattern
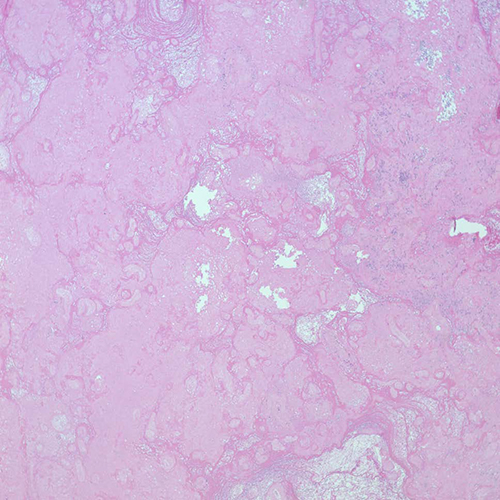
Figure 4 (H&E, 2x) The normal architecture of the placenta is distorted and there is massive fibrin deposition.
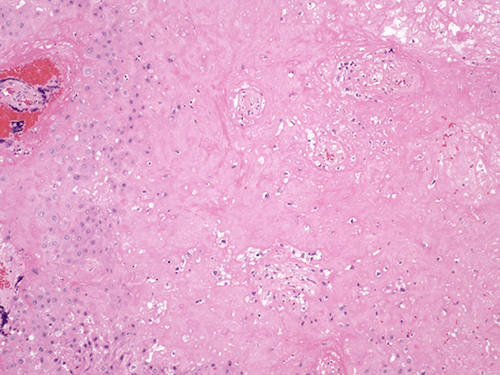
Figure 5 (H&E, 10x) Chorionic villi that are atrophic, widely separated and surrounded by cloaks of fibrin/fibrinoid material.
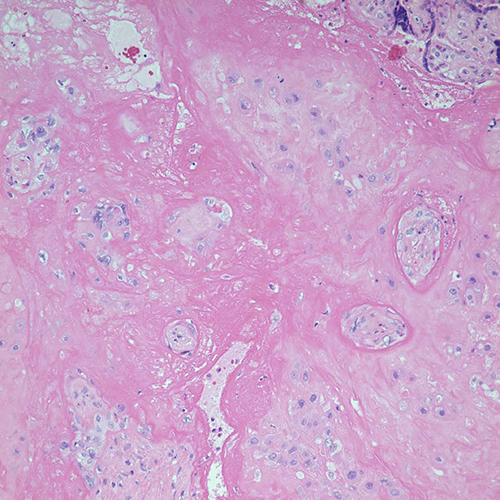
Figure 6 (H&E, 20x) Chorionic villi that are atrophic, widely separated and surrounded by cloaks of fibrin/fibrinoid material associated with trophoblast proliferation
Diagnosis
Massive perivillous fibrin deposition
Discussion
Maternal floor infarction (MFI) and massive perivillous fibrin deposition (MPVFD) are rare and closely related placental lesions of unclear etiology. These placental lesions are associated with high perinatal morbidity, mortality, and recurrence risks. The incidence rate is estimated to be 0.025-0.5% of deliveries. On prenatal ultrasound, findings include fetal intrauterine growth restriction, oligohydramnios, and dense hyperechoic placenta. MPFVD is associated with a high recurrence rate 12-78%.
The diagnosis should be suspected when there are gross findings of a diffusely firm placenta with a maternal surface that is diffusely pale yellow-white and serial sections show deposition of granular fibrin/fibrinoid material extending upward towards the chorionic plate in a matted or trabecular network. The diagnosis is confirmed by microscopic examination which is characterized by chorionic villi that are atrophic, widely separated and surrounded by cloaks of fibrin/fibrinoid material. The perivillous deposition is greatest in the basal zone with variable penetration into the midchorionic and subchorionic zones. Syncytiotrophoblastic damage is the first detectable villous change, but whether it is the initiating or merely an early event remains unknown.
Intermediate trophoblast proliferation is usually seen. Decidual arteriopathy, which is typically seen in hypertensive disorders, is usually absent in MPVFD. MPVFD may coexist with chronic villositis. It has been suggested that MPVFD should be subclassified into classic, borderline, and transmural. The classic pattern is characterized by basal villous encasement by fibrin/fibrinoid along the entire maternal floor and of 3 mm or greater thickness on at least one slide. Borderline MPVFD refers to involvement of 25-50% of villi on at least one slide in transmural or nearly transmural distribution. Transmural MPVFD refers to transmural perivillous fibrin/fibrinoid extension with encasement of 50% or greater of villi on at least one slide.
The differential diagnosis includes physiologic perivillous fibrin/fibrinoid deposition, chorionic villous ischemia and infarction, fetal vascular malperfusion (FVM), diffuse chorionic villitis of unknown etiology (diffuse VUE), and massive chorionic histiocytic noninfectious intervillositis (MCI). In villous infarction the villi show signs of necrosis and fibrin surrounds the villi but the intervillous space is collapsed unlike in MPVFD where it is expanded. FVM is characterized by clusters or large areas of avascular, involuted appearing distal terminal villi, due to intermittent, or sudden obstruction of blood flow within the villous tree. FVM lesions typically do not show perivillous fibrin/fibrinoid deposition although long standing lesions may have foci of avascular villi with fibrinoid encasement. Diffuse VUE are high-grade destructive lymphohistiocytic villitides usually occurring in mid to late third trimester placentas. These lesions can feature perivillous fibrin/fibrinoid deposition around individual or clusters of inflamed chorionic villi. MPVFD lesions can show scant foci of chorionic villitis and do not demonstrate chorionic stem vessel vasculitis or thrombosis of FVM. MCI is characterized by diffuse mononuclear infiltrates consisting of lymphocytes, monocytes and histocytes within the intervillous space, absence of villitis and may demonstrate mild villous and intervillous fibrin/fibrinoid deposition.
While the etiology of MPVFD is unknown, it is more commonly seen in women with antiphospholipid antibody syndrome (APL) and other autoimmune disorders with or without thrombophilia. Microscopically the findings in MPVFD suggest either direct damage to the syncytiotrophoblasts or disturbances in the balance of dually derived intervillous procoagulant/anticoagulant factors in the maintenance of fluid state of the blood within the maternal space.
References
- Chen A and Roberts DJ. Placental pathologic lesions with a significant recurrence risk - what not to miss! APMS. 2017. 126:589-601.
- Weber MA et al. Co-occurrence of Massive Perivillous Fibrin Deposition and Chronic Intervillositis: Case Report. 2006. Pediatric and Developmental Pathology. 2006. 9:234-8.
- Faye-Petersen OM and Ernst LM. Maternal Floor Infarction and Massive Perivillous Fibrin Deposition. Surgical Pathology. 2013. 6:101-14.
- Chaiworapongsa T et al. Pravastatin to prevent recurrent fetal death in massive perivillous fibrin deposition. Journal of Maternal-Fetal & Neonatal Medicine. 2015. Early Online. 1-8.
- Abdulghani S et al. Growth Restriction, Osteopenia, Placental Massive Perivillous Fibrin Deposition With (or Without) Intervillous Histiocytes and Renal Tubular Dysgenesis - An Emerging Complex. Ped and Dev Path. 2018. 21(1):91-94.
Have Questions or Need Help?
If you have questions or would like to learn more about the Anatomic and Clinical Pathology Residency Program at Cedars-Sinai, please call or send a message to Academic Program Coordinator, LeeTanya Marion-Murray.
Department of Pathology and Laboratory Medicine
8700 Beverly Blvd., Room 8709
Los Angeles, CA 90048-1804
