December 2019 Case
Authors
Corey Chang, MD, PhD (PGY-2), Kevin Waters, MD, PhD (Faculty)
Subject: Gastrointestinal pathology
Clinical History
A woman in her 30s with a history of bilateral invasive ductal carcinoma with focal lobular features status post bilateral mastectomy underwent genetic panel testing which revealed a germline CDH1 mutation. Screening endoscopy demonstrated a focus of signet-ring cell adenocarcinoma involving and confined to the lamina propria within oxyntic mucosa in one of six tissue fragments of randomly sampled transition zone. She subsequently underwent elective prophylactic total gastrectomy with lymphadenectomy and was found to have multiple foci of signet-ring cell adenocarcinoma confined within the superficial lamina propria (pT1aN0).
Figures
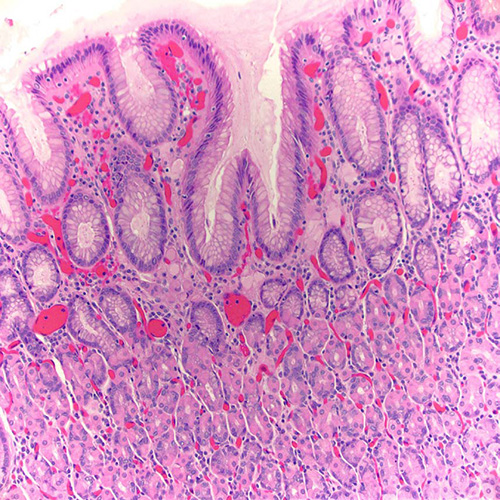
Figure 1. (H&E, 100x): Permanent section
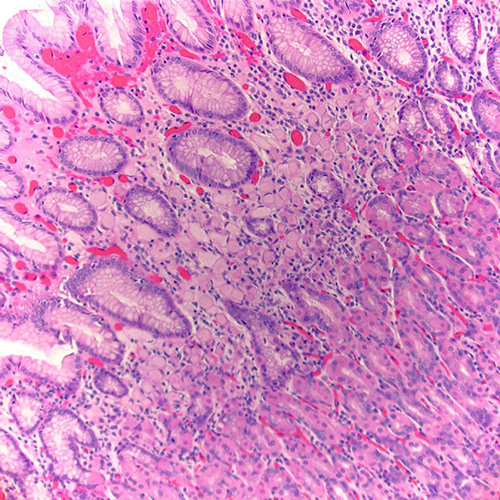
Figure 2. (H&E, 200x): Permanent section
Diagnosis
Signet ring gastric adenocarcinoma, diffuse type
Discussion
Gastric cancer is the third most common cause of cancer-related death in the world and remains difficult to cure primarily because most patients present with advanced disease. Gastric cancer is a heterogeneous disease and hereditary cancer syndromes constitute <3% of cases. The Lauren's classification scheme divides/classifies gastric cancers into histologic subtypes: intestinal, diffuse, mixed and indeterminate. The intestinal subtype is associated with gastric atrophy and intestinal metaplasia, gland formation (differentiated) and exhibits hematogenous spread. The diffuse subtype is associated with poorly differentiated signet ring cells and typically undergoes transmural/lymphatic spread. Environmental factors are a larger component the intestinal subtype, while genetic factors, including germline familial cases, play a relatively larger role in the diffuse subtype which is associated with decreased E-cadherin expression. Microscopically, the signet ring cell has a characteristic morphology with intracellular mucin with an eccentrically displaced nucleus. T staging is based on depth of invasion: T1a refers to invasion to the lamina propria or muscularis mucosae, T1b refers to invasion to the submucosa, T2 refers to invasion into the muscularis propria, T3 refers to penetration of the subserosal connective tissue without invasion of the visceral peritoneum or adjacent structures, T4a refers to invasion of the serosa (visceral peritoneum) and T4b refers to invasion to adjacent structures/organs.
Hereditary diffuse gastric cancer (HDGC) is defined as an autosomal dominant cancer susceptibility syndrome characterized by diffuse (signet ring cell) gastric cancers at a young age. E-cadherin (CDH1) is a glycophorin protein (tumor suppressor gene) which mediates cell-cell adhesion in a calcium dependent manner. Interaction of CDH1 with the actin cytoskeleton is required for membrane deformation processes (i.e., endocytosis, exocytosis, autophagy, receptor/channel recycling) thereby playing an important role in maintenance and homeostasis of the epithelium in adult tissue. Reduction or complete absence of E-cadherin is associated with loss of epithelial morphology and increased invasiveness through the epithelial-mesenchymal transition. It is estimated that up to 30% of families with HDGC harbor germline CDH1 mutations. Unlike sporadic diffuse gastric cancer, HDGC individuals inherit a mutated allele and subsequently undergo loss of heterozygosity of the remaining CDH1 allele (most commonly through CDH1 promoter hypermethylation). Genetic testing for CDH1 mutations should be considered using the following criteria: 1) >2 documented cases of diffuse gastric cancer in first- or second-degree relatives, with >1 diagnosed before age 50, 2) >3 cases of diffuse gastric cancer in first- or second- degree relatives, independent of age of onset, 3) diffuse gastric cancer diagnosed before age 40 years without a family history and 4) personal or family history of diffuse gastric cancer and lobular breast cancer, one diagnosed before age 50 years. Recent studies suggest that HDGC progresses through several stages and that even when the tumor invades the lamina propria, it may stay indolent for a long time.
Prophylactic total gastrectomy (PTG) is recommended between the ages of 18 and 40 for CDH1 mutations carriers. A baseline endoscopy is indicated prior to prophylactic total gastrectomy, although multiple series have shown that early lesions are not evident endoscopically. Intraoperative frozen sections should be performed to verify that the proximal margin contains esophageal squamous mucosa and the distal margin contains duodenal mucosa to ensure complete removal of gastric tissue. While not recommended, PTG may be considered in certain individuals prior to age 18, especially those with family members diagnosed with gastric cancer before 25 years of age. Women with CDH1 mutations are at increased risk for breast cancer (lobular carcinoma). The current screening recommendation is for annual mammogram with consideration of tomosynthesis and breast MRI with contrast starting at 30 years of age. Risk reducing mastectomy can be performed based on family history, but the evidence is currently insufficient for a strong recommendation.
References
- Figueiredo J et al. Clinical spectrum and pleiotropic nature of CDH1 germline mutations. J Med Genet (2019). 56:199-208.
- Luo W et al. CDH1 Gene and Hereditary Diffuse Gastric Cancer Syndrome: Molecular and Histological Alterations and Implications for Diagnosis and Treatment. Front In Pharm (2018). 9;1421.
- McLean MH and El-Omar EM. Genetics of gastric cancer. Nature Rev (2014). 11:664-74.
- NCCN Guidelines Version 3.2019. Gastric Cancer. Principles of Genetic Risk Assessment for Gastric Cancer.
- NCCN Guidelines Version 3.2019. Genetic/Familial High-Risk Assessment: Breast and Ovarian. Breast and Ovarian Management Based on Genetic Test Results.
Have Questions or Need Help?
If you have questions or would like to learn more about the Anatomic and Clinical Pathology Residency Program at Cedars-Sinai, please call or send a message to Academic Program Coordinator, LeeTanya Marion-Murray.
Department of Pathology and Laboratory Medicine
8700 Beverly Blvd., Room 8709
Los Angeles, CA 90048-1804
