August 2019 Case
Authors
Brian Cox, MD, MAS (PGY-3); Stacey Kim (Faculty)
Subject: Gastrointestinal Pathology
Clinical History
76-year-old female with diabetes mellitus type 2, hypertension, and coronary artery disease presented to the emergency department with a one-month history of right lower quadrant abdominal pain that worsened over the last three days. At the time of presentation, she was afebrile but endorsed nausea and intermittent vomiting. A subsequent CT scan showed evidence of an acute appendicitis and appendicolith with no evidence perforation. She was urgently referred for laparoscopic resection. Intraoperatively, the appendix was thickened, inflamed, and adherent to both the retroperitoneum and cecum. Grossly, the appendiceal serosa was significant for multiple sub-centimeter excrescences and a 0.2 cm perforation at the mid appendix. Pathology confirmed a low-grade goblet cell adenocarcinoma at the proximal resection margin. The tumor invaded the muscularis propria and subserosa but did not extend beyond the serosa. Given the proximity of the tumor to the resection margin, the patient underwent a right hemicolectomy. No residual tumor or lymph node invasion was identified.
Figures
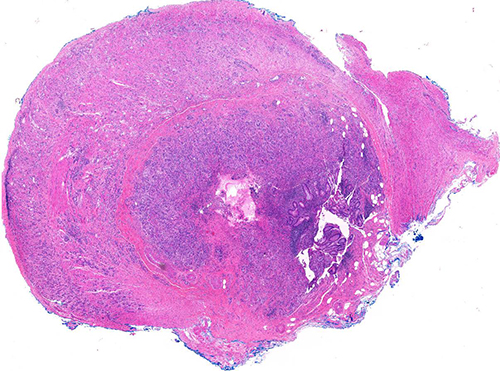
Figure 1 (H&E, 2x)
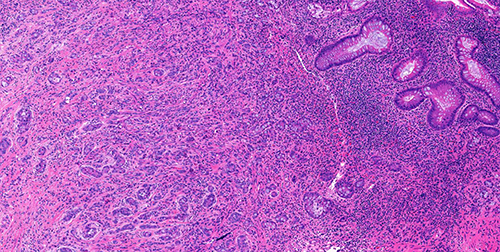
Figure 2 (H&E, 20x)
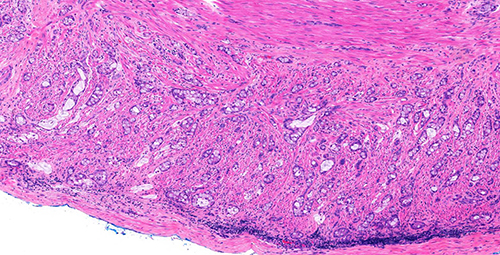
Figure 3 (H&E, 20x)
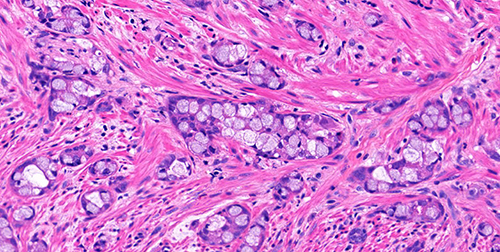
Figure 4 (H&E, 40x)
Diagnosis
Goblet cell adenocarcinoma, low grade (G1)
Discussion
Goblet cell adenocarcinoma (GCA) is a rare and distinctive neoplasm of the appendix that accounts for less than5% of all appendiceal neoplasms. The tumor is thought to originate from pluripotent intestinal crypt base stem cells which differentiate into both glandular and neuroendocrine components. The amalgamation produces mixed histologic patterns that are generally characterized by discrete nests of goblet cells admixed with neuroendocrine secretory granules and occasional Paneth cells. The tumor's rarity and variable histology has resulted in a litany of synonymous nomenclature including: goblet cell carcinoid, mucinous carcinoid tumor, mixed crypt cell carcinoma, adenocarcinoid-goblet cell type, microglandular goblet cell carcinoma, and adenocarcinoma ex-goblet cell carcinoid.
GCAs are almost exclusively encountered in the appendix. In most cases, tumor cells originate from the deep lamina propria and cause concentric appendiceal wall thickening. As in this case, patients typically present with acute appendicitis and the tumor is identified incidentally after appendectomy. When confined to the appendix, these tumors are considered low-grade with a prognosis better than appendiceal adenocarcinoma but worse than classic neuroendocrine tumors of the appendix. Although aggressive GCAs demonstrate preferential transcoelomic dissemination with metastasis to gynecologic organs, the appendix-confined well-differentiated examples are reportedly curable by appendectomy. However, right hemicolectomy is recommended for even low grade GCAs as approximately half of patients who undergo right hemicolectomy are reported to have residual disease.
Over the last 30 years, multiple authors have attempted to characterize which histologic features are associated with poor clinical prognosis. One of the first studies conducted by Pham et al. (2006) showed disease-specific 5-year survival for AJCC stages I, II, III, and IV were 100%. 76%, 22%, and 14%, respectively. The authors noted that tumors amenable to surgical resection were prognostically favorable as GCAs showed lackluster response to traditional adenocarcinoma chemotherapy. Since GCAs appear to manifest and behave as an adenocarcinoma, Tang et al. (2008) compared 63 cases of GCAs and stratified cases according to the presence and differentiation of adenocarcinoma. Using a three-tiered system, they found that GCAs with a poorly differentiated adenocarcinoma component were associated with worse prognosis. Taggert et al. (2014) attempted to revise this grading system by assessing the percent of adenocarcinoma present in each GCA. They stratified a cohort of 142 tumors into four groups and concluded that both tumor stage and an adenocarcinoma component >50% represent independent poor prognostic factors. However, the reproducibility of these grading systems was source of consternation and confusion for both pathologists and clinicians. This led Yozu et al. (2018) to not only suggest that all 'goblet cell carcinoids' be renamed goblet cell adenocarcinomas but also that all GCAs be graded as either low or high grade based on several specific criteria. At the time of this report, no official consensus has been reached.
In summary, this case highlights a rare but important appendiceal neoplasm. The case presentation, clinical management, and favorable prognosis illustrate the low-grade nature of the tumor when identified at an early stage. In contrast, higher grade GCAs may present as a gynecological metastasis or with disseminated disease. Although this clinical presentation raises the possibility of an ovarian or mucinous appendiceal neoplasm, the lowpower concentric infiltration of goblet cells is distinct on histology.
References
- Burke AP, Sobin LH, Federspiel BH, et al. Goblet cell carcinoids and related tumors of the vermiform appendix. Am J Clin Pathol. 1990;94:27–35
- Pham TH, Wolff B, Abraham SC, Drelichman E. Surgical and chemotherapy treatment outcomes of goblet cell carcinoid: a tertiary cancer center experience. Ann Surg Oncol 2006;13:370.
- Tang LH, Shia J, Soslow RA, et al. Pathologic classification and clinical behavior of the spectrum of goblet cell carcinoid tumors of the appendix. Am J Surg Pathol. 2008;32:1429–1443
- Taggart MW, Abraham SC, Overman MJ, et al. Goblet cell carcinoid tumor, mixed goblet cell carcinoidadenocarcinoma, and adenocarcinoma of the appendix: comparison of clinicopathologic features and prognosis. Arch Pathol Lab Med. 2015;139:782–790.
- Reid MD, Basturk O, Shaib WL, et al. Adenocarcinoma ex-goblet cell carcinoid (appendiceal-type crypt cell adenocarcinoma) is a morphologically distinct entity with highly aggressive behavior and frequent association with peritoneal/intra-abdominal dissemination: an analysis of 77 cases. Mod Pathol. 2016;29(10):1243–1253.
- Wang YT, Li YR, Ke TY. Adenocarcinoma Ex Goblet Cell Carcinoid of Appendix: Two Case Reports. Case Rep Pathol. 2017;2017:5930978.
- Yozu M, Johncillia M, Srivastava A, et al. Histologic and Outcome Study Supports Reclassifying Appendiceal Goblet Cell Carcinoids as Goblet Cell Adenocarcinomas, and Grading and Staging Similarly to Colonic Adenocarcinomas. Am J Surg Path. 2018;42(7):898-910.
Have Questions or Need Help?
If you have questions or would like to learn more about the Anatomic and Clinical Pathology Residency Program at Cedars-Sinai, please call or send a message to Academic Program Coordinator, LeeTanya Marion-Murray.
Department of Pathology and Laboratory Medicine
8700 Beverly Blvd., Room 8709
Los Angeles, CA 90048-1804
